Our Technology
The Organoid Sorter: A Platform for 3D Biology & New Approach Methodologies (NAMs)
The Rise of 3D Biology and New Approach Methodologies (NAMs)
Three-dimensional (3D) cell culture models, including organoids and spheroids, are reshaping how researchers study human biology. By capturing tissue-like structure, multicellular organization, and functional complexity, these systems offer the promise to provide more physiologically relevant insights than traditional 2D cell cultures. What began as specialized models and low-yield culture techniques are becoming foundational to disease modeling, drug discovery, and translational research.
At the same time, global regulatory and scientific communities are advancing New Approach Methodologies (NAMs) – frameworks designed to reduce reliance on animal testing while improving the human-relevance of the models we use to make decisions about drug efficacy and safety. Organoids, spheroids, and other multicellular 3D systems are increasingly explored as potential NAMs, along with small model organisms like Caenorhabditis elegans (C. elegans) and Danio rerio (zebrafish).
As momentum builds, the field is encountering a new and critical challenge: how to scale, standardize, and operationalize complex 3D biology. For organoids and spheroids to be established as NAMs, researchers must be able to:
- Work with intact 3D structures, not just dissociated cells
- Generate reproducible measurements across experiments and sites
- Increase throughput without compromising biological integrity
- Automate handling of large, fragile, and heterogeneous samples
Meanwhile, many laboratories are still defining what quality, phenotypes, and functional readouts mean for these emerging models. Analytical tools must therefore be flexible, enabling both routine measurement and exploratory discovery as the field matures.
Purpose-built instrumentation for flow-based analysis, brightfield imaging, bulk and multi-well dispensing, and automated sampling provides the foundation for moving 3D biology from small-scale, manual workflows towards standardized, scalable processes. By enabling researchers to analyze, quantify, sort, and handle intact organoids and spheroids efficiently, these technologies help translate the promise of 3D models into practical, reproducible NAM applications.
Analysis of Intact Organoids by Flow Cytometry
Traditional flow cytometry is a cornerstone of cell biology. For decades, researchers have used flow cytometry to rapidly measure physical and fluorescent properties of thousands of individual cells in suspension. Parameters such as size, granularity, and marker expression are captured as each cell passes through a laser interrogation point, enabling high-throughput, quantitative analysis of complex cell populations.
However, conventional flow cytometry requires 3D cellular structures to be dissociated into single cells. For 3D models such as organoids and spheroids, this step introduces significant limitations. Dissociation adds processing time, increases variability, and disrupts the very architecture that gives these models their biological relevance. Spatial organization, multicellular structure, and morphology are lost when organoids are reduced to single-cell suspensions.
Union Biometrica, Inc.’s platform extends the principles of flow cytometry to intact 3D biological structures. Instead of analyzing only single cells, researchers can measure whole organoids and spheroids in flow, collecting quantitative data from complex multicellular models in a manner analogous to traditional flow cytometry and FACS workflows.
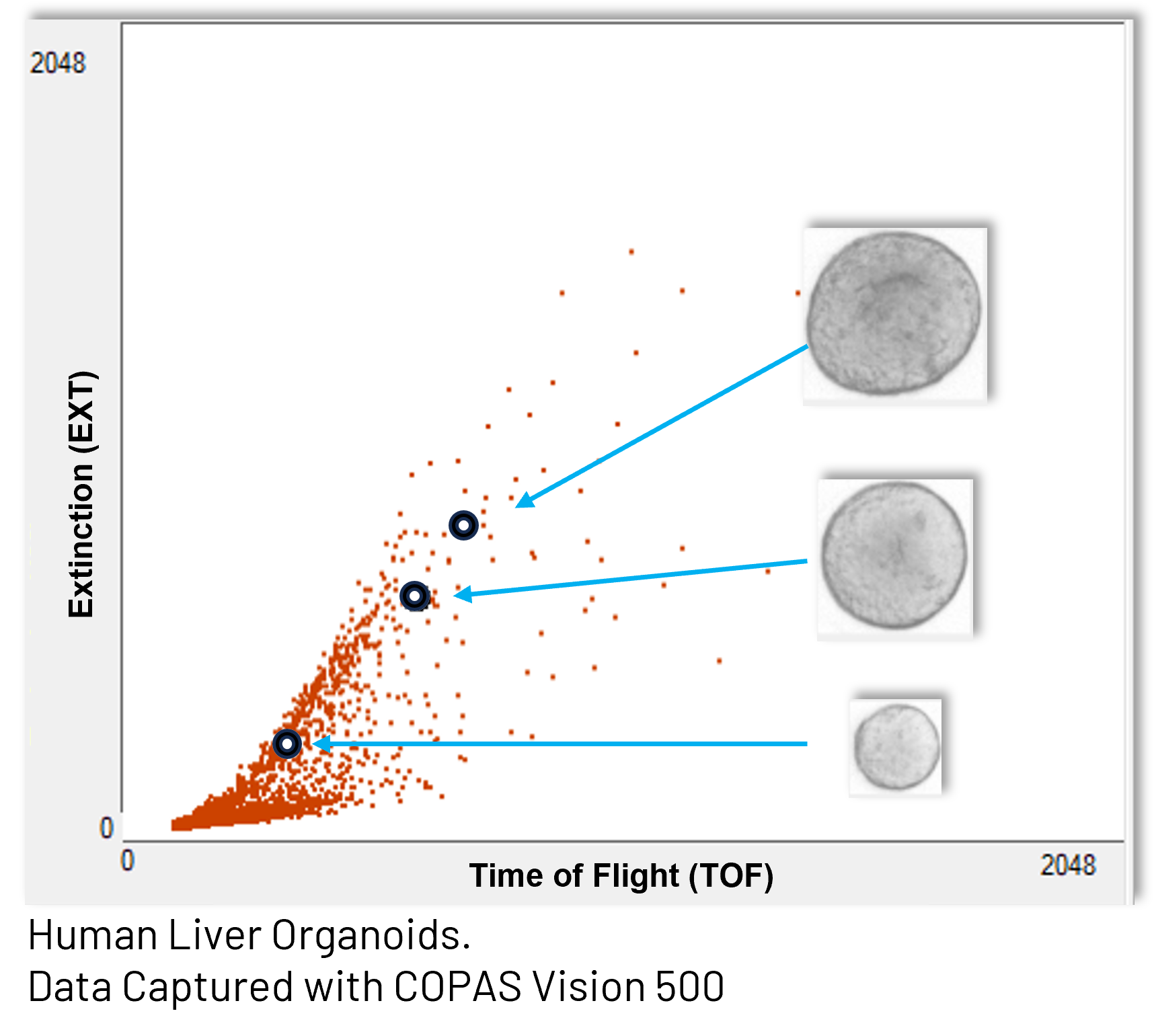
Our systems support a wide size range, from small multicellular aggregates to organoids up to 1.5 mm in diameter. A range of flow cell options – including 250, 500, 1000, and 2000 micron sizes – enables analysis of heterogeneous samples containing structures of various sizes and morphologies.
As intact organoids pass through the flow cell, multiple parameters can be measured, including:
- Time of Flight (TOF) as an indicator of size of the organoid
- Extinction (EXT) as an indicator of the optical density of the organoid
- Forward Scatter and Side Scatter indicate the light scattering off of the organoid
- Fluorescence signals from reports, stains, and labeled populations of cells in the organoid
These measurements allow researchers to characterize populations of organoids just as they would cell populations – using dot plots, gating strategies, and multi-parametric analysis – while preserving the biological context of the 3D structure.
By linking quantitative flow parameters with brightfield imagery, researchers can directly connect signal patterns to organoid morphology. This provides a powerful way to explore heterogeneity within organoid cultures and to define subpopulations based on size, structure, or developmental state. Fluorescent reporters and stains add another layer of information, enabling measurement of gene expression, viability, or cell-type composition within intact organoids.
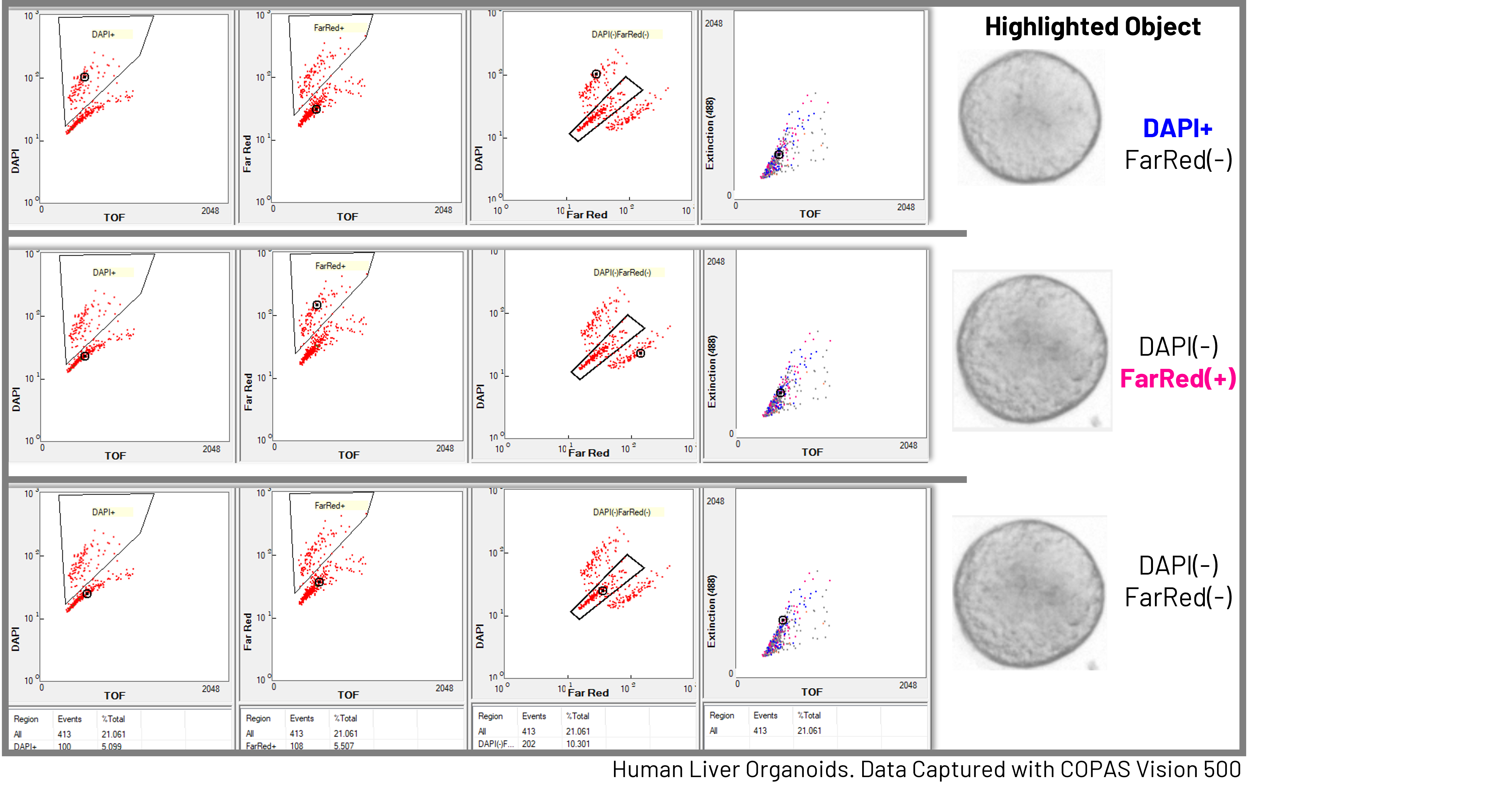
Together, these capabilities transform flow cytometry from a single-cell technique into a tool for population-level analysis of intact 3D biological models.
Profiler – Beyond the Integral Signal
In conventional flow cytometry, fluorescence and light scatter measurements are typically captured as a single integrated value, effectively the area under the signal curve as the cell passes through the laser. For small, relatively uniform cells, such as blood cells, this provides sufficient information to distinguish populations.
Larger and more structurally complex objects, such as spheroids and organoids, contain more spatial information than may be represented by a single integral signal. Differences in how cells are organized within a 3D structure can be biologically meaningful but invisible to traditional cytometry measurements.
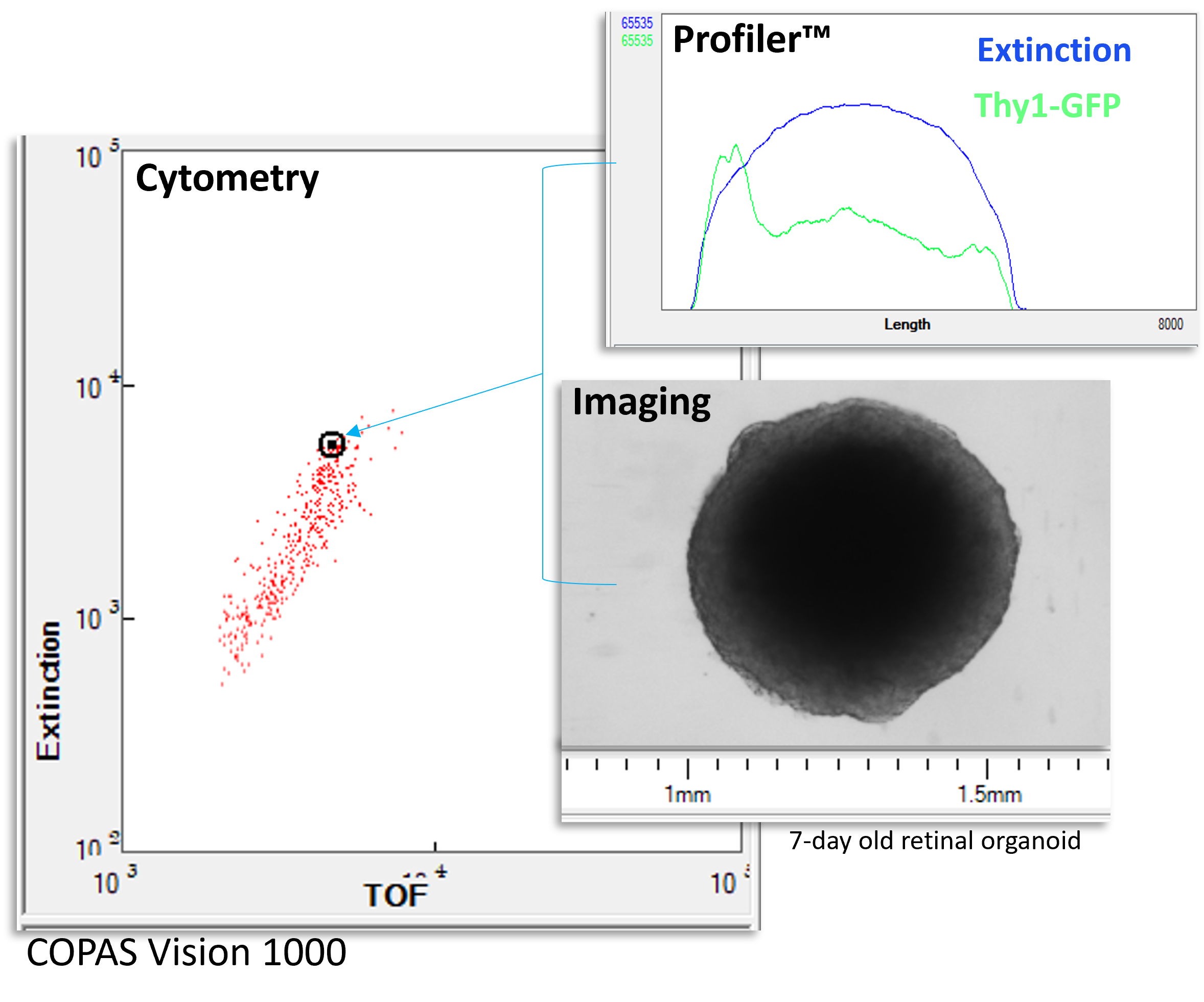
Profiler technology extends flow-based analysis by capturing signal profiles across the length of each object as it passes through the interrogation region. Instead of one integral value, researchers obtain a spatially resolved signal that reflects how fluorescence or optical properties vary within the structure.
This enables new types of questions to be addressed, such as:
- How are different cell types arranged within a spheroid?
- Does a reporter localize to the outer layers or internal regions of an organoid?
- How does structural organization change during development or in response to treatment?
By moving beyond integral measurements, Profiler analysis brings spatial context into flow-based workflows, bridging the gap between quantitative cytometry and structural biology.
Analysis and Imaging – Imaging Flow Cytometry
Organoids and spheroids are inherently complex. No single measurement can fully describe their size, structure, composition, or developmental state. Combining multiple, complementary data types is a way to provide meaningful characterization of 3D cultures.
The COPAS Vision™ brings together population-level cytometry data, object-level Profiler data, and in-flow brightfield imaging into a single, integrated workflow. As each organoid passes through the flow cell, optical measurements are collected alongside brightfield images, creating a multidimensional dataset that links quantitative signals directly to visible morphology.
This orthogonal data collection is especially powerful for researchers working with emerging 3D models, where analytical standards are still being established. Cytometry parameters provide high-throughput, quantitative population analysis, while imaging supplies the visual context that biologists rely on when evaluating cultures under a microscope.
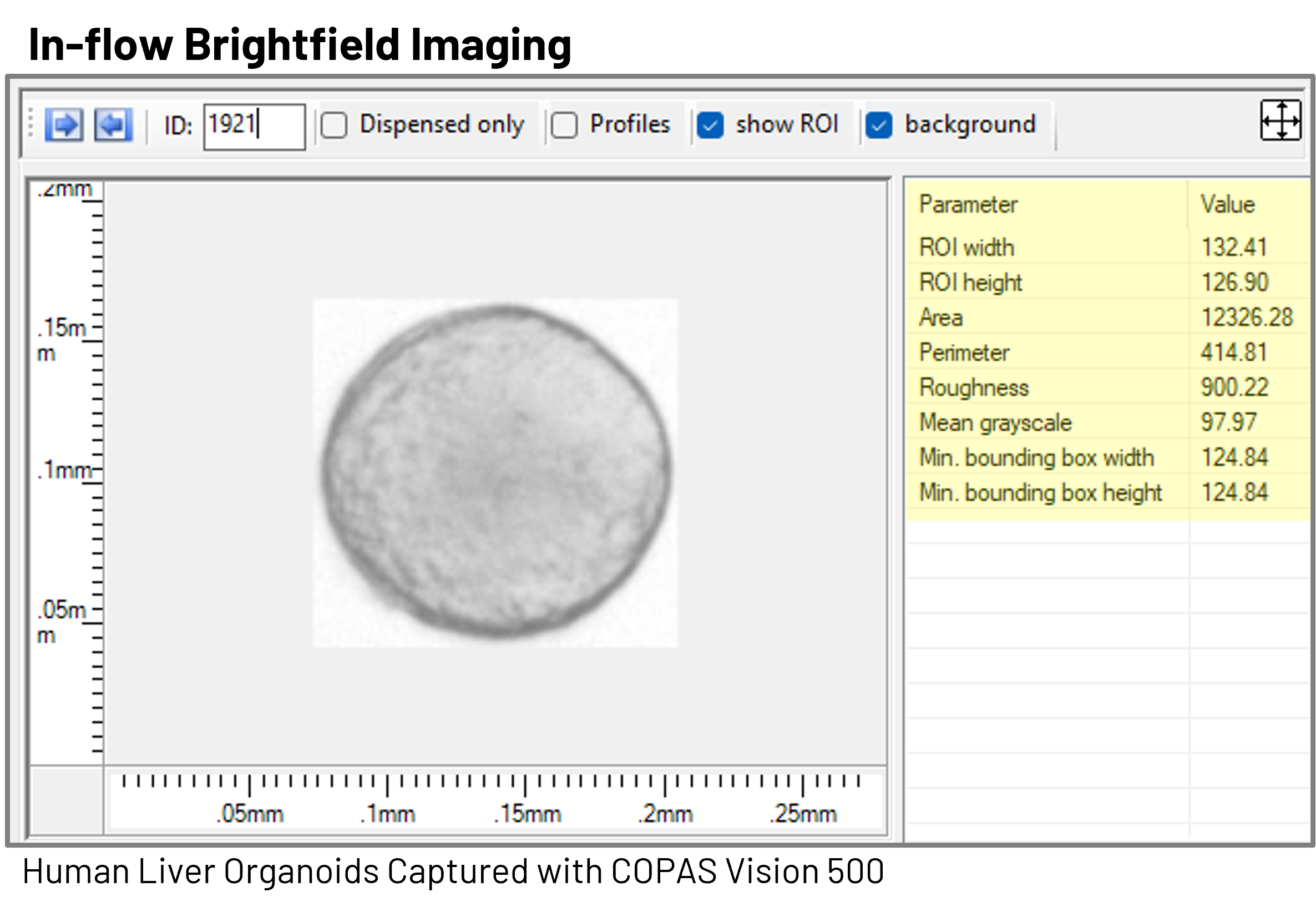
By capturing brightfield images in flow, researchers can confirm that the populations defined by cytometry parameters correspond to the expected organoid morphologies. This helps ensure that gating strategies are selecting biologically relevant structures rather than debris, aggregates, or outliers. In this way, imaging acts as a bridge between traditional microscopy-based assessment and flow-based measurement.
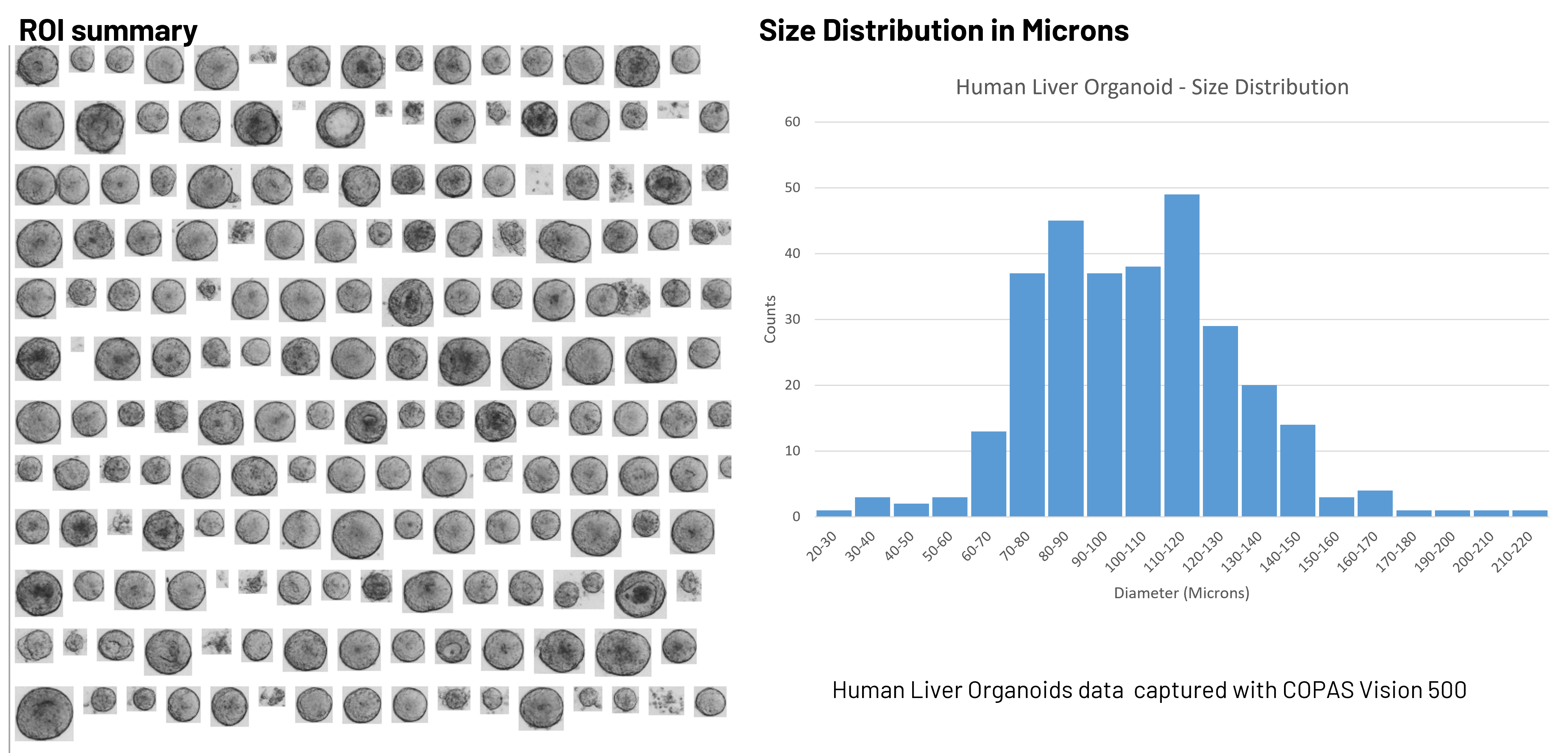
Beyond visual confirmation, image-derived measurements provide additional quantitative descriptors of organoids. These can include:
- Size measurements in microns derived directly from image dimensions
- Texture metrics that reflect structural integrity
- Mean grayscale features associated with density or internal organization
These parameters can be used alongside optical and fluorescence data to better distinguish subpopulations within heterogeneous 3D cultures.
Maintaining an image record linked to each flow or sorting dataset is particularly valuable during method development and model optimization. As researchers refine culture conditions or establish new organoid systems, archived images allow retrospective analysis – enabling new features or classification strategies to be applied to previously acquired data without repeating experiments.
By integrating Cytometry, Profiler, and Imaging, the COPAS Vision provides a comprehensive approach to characterizing 3D biological models – combining the statistical power of flow with the interpretability of microscopy.
Counting and Sorting – Batch Analysis and Custom Sorting in Bulk and Multi-Well Plates
As 3D culture systems become more widely adopted, researchers need ways not only to characterize organoids, but also to quantify and physically handle them in a controlled, reproducible manner. Flow-based analysis provides a natural extension of familiar cytometry principles to meet this need.
Using the same population-level framework that cell biologists have long used for single-cell suspensions, intact organoids can be counted, sized, and classified in flow. Parameters such as TOF, Extinction, Scatter, and Fluorescence allow heterogeneous cultures to be broken down into defined subpopulations, revealing distributions of organoid size, composition, or reporter expression across an entire sample.
Because these measurements are collected event by event, researchers can generate true population statistics for organoid cultures, not just estimates from manual microscopy. This makes it straightforward to monitor culture consistency, compare experimental conditions, or define quality metrics as 3D models are scaled for screening or NAM workflows.
Beyond analysis, these same measurements can be used to gently sort and dispense organoids based on user-defined criteria.
Sorting and dispensing are highly flexible and can be performed in:
- Bulk formats, for collecting selected organoid populations into 50mL conical tubes or other containers
- Multi-well plates, including standard plate formats (24-, 48-, 96-, and 384-well plates)
Union Biometrica, Inc.'s large particle flow cytometry systems are compatible with standard laboratory multi-well plate formats and can be configured to match existing workflows, making it possible to integrate organoid selection and distribution directly into established screening or assay pipelines.
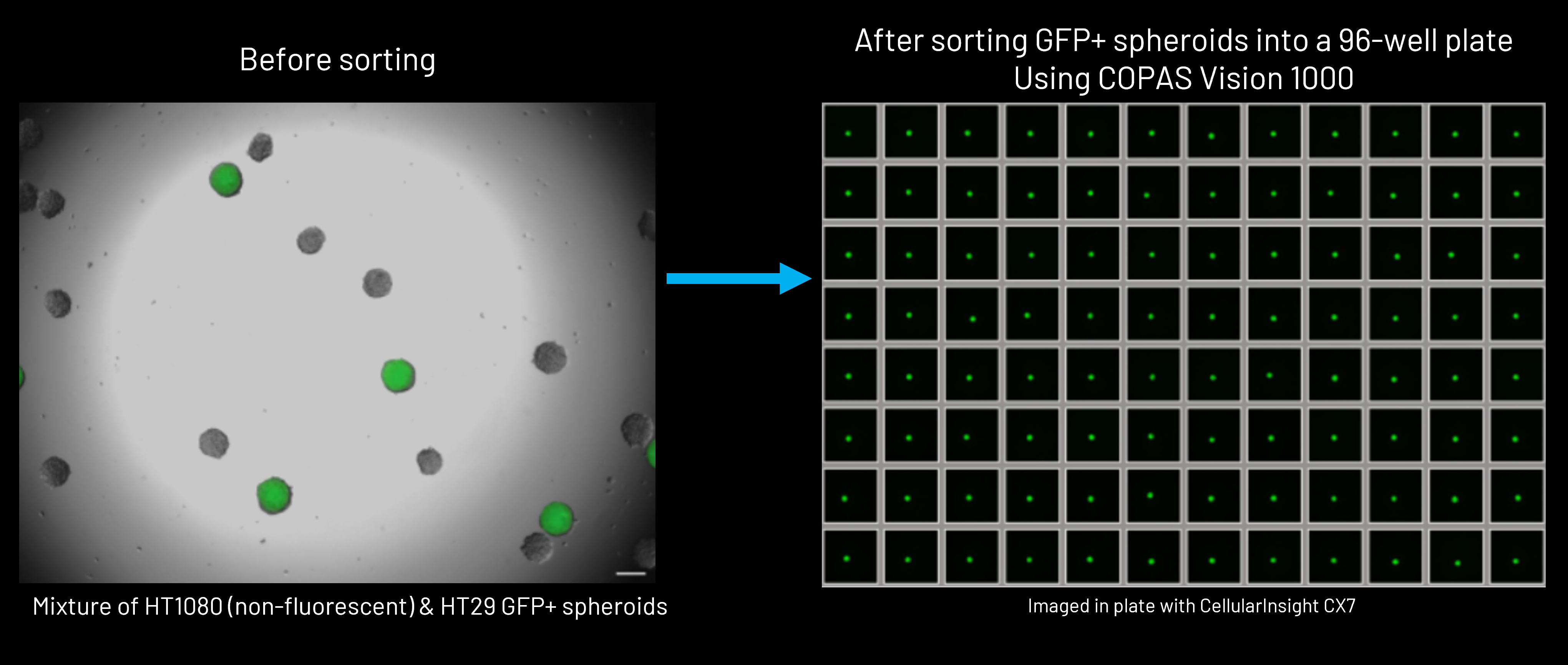
This ability to move seamlessly from measurement to controlled physical handling transforms flow cytometry from a purely analytical tool into a practical platform for building reproducible 3D biology workflows. Researchers can not only define organoid populations, but also isolate, distribute, and standardize them at the scales demanded of modern screening and experimental design.
As 3D biological models move into routine use for screening, disease modeling, and NAM-driven applications, manual handling quickly becomes a limiting factor. Variability in plating, transfer, and processing can obscure true biological signals and make standardization difficult across experiments or laboratories. To address this, large particle flow cytometry/FACS platforms can be configured as integrated automation systems for 3D biology workflows.
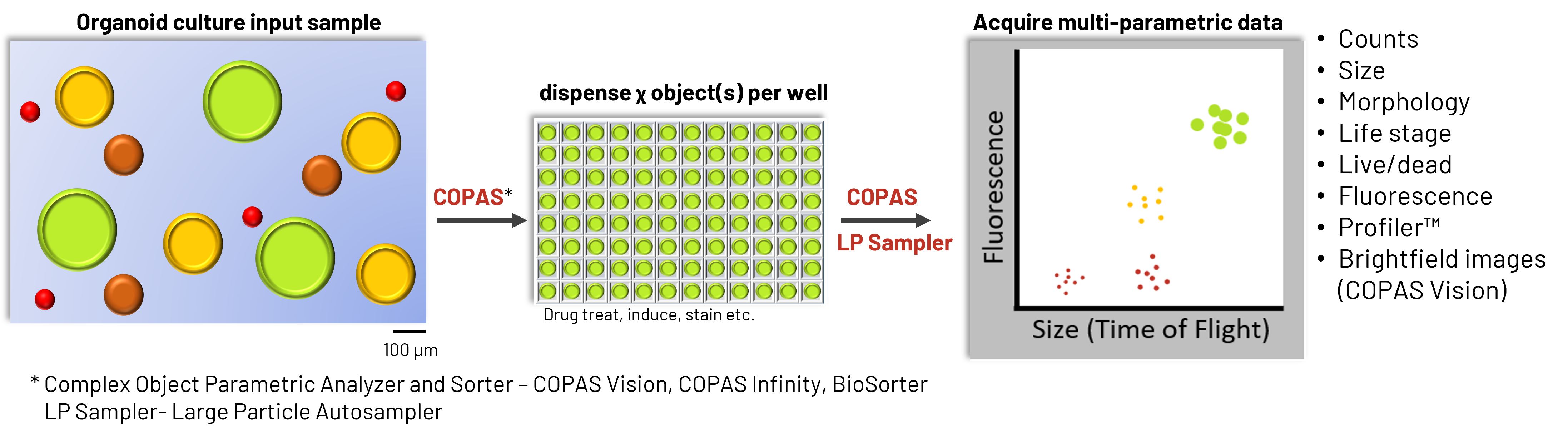
Rather than functioning as standalone instruments, these systems operate as a coordinated platform for the analysis, counting, sorting, and dispensing of intact organoids and spheroids.
Automated Handling of Intact 3D Models
The integration of an LP Sampler enables automated sample loading of intact organoid samples, reducing manual intervention and improving consistency across runs. This is particularly valuable when working with fragile or heterogeneous 3D cultures where gentle, reproducible handling is critical for maintaining biological integrity.
Automation at the sampling stage supports:
- Higher-throughput analysis of organoid cultures
- Improved reproducibility between samples and experiments
- Reduced operator-dependent variability
By combining measurement and controlled distribution in a single workflow, researchers can move from heterogeneous culture to structured experimental layout with minimal manual handling. This supports the transition of organoid models from exploratory research tools to standardized assay platforms.
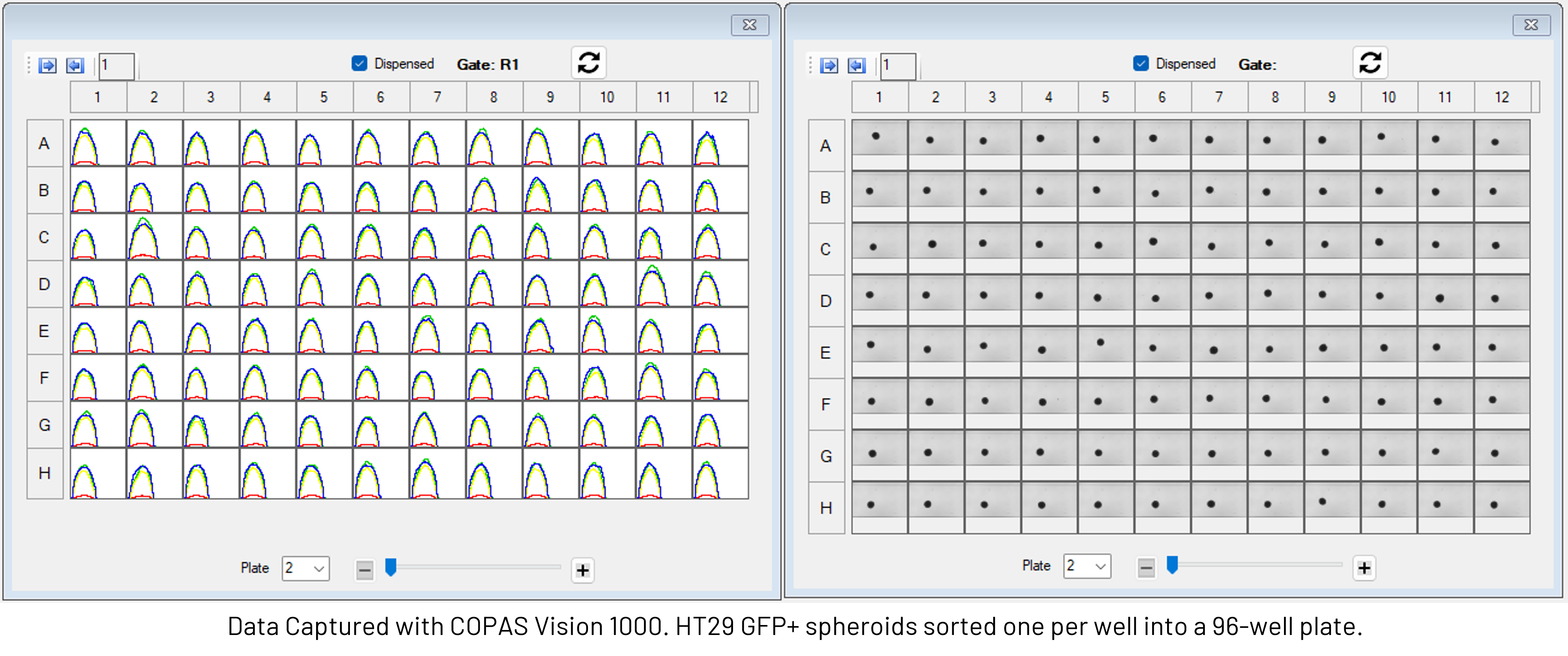
Index Sorting is a feature of the FlowPilot software that makes it is easier to link the full phenotype of every sorted object to the well location in the plate it was dispensed into. Multi-parametric data captured from each object are associated back to the plate layout, enabling researchers to trace the phenotype of an organoid or spheroid to its downstream fate. This traceability ensures experiments are reproducible, well-documented, and auditable, supporting both robust bioprocess development and compliance.
Designed for Biosafety and Sterility
The COPAS Infinity and COPAS Vision systems are designed for operation within a standard Class II biosafety cabinets, supporting safe workflows when using human-derived or potentially hazardous biological materials. Cabinet integration helps protect:
- Personnel, by containing aerosols
- Samples, through HEPA-filtered airflow
- The laboratory environment, via controlled exhaust
In addition, the enclosed sorting chamber provides an additional layer of containment, and onboard UV sterilization supports routine decontamination of internal components between runs. Together, these features help align automated 3D biology workflows with institutional biosafety requirements.

By combining autosampling, imaging, cytometry, and flexible dispensing, this platform approach enables researchers to build end-to-end workflows for 3D biology – from culture characterization through standardized experimental setup. As NAM initiatives drive demand for scalable, human-relevant models, our automated large particle systems provide the infrastructure needed to move complex 3D cultures into standard, industry-scale use.

